Epidemiology & Biostatistics
Genenerating the evidence needed to support clinical, regulatory, and safety decisions.
BlueRidge Life Sciences’ Epidemiology and Biostatistics experts support life sciences organizations through evidence strategy and planning, study design, execution, interpretation, and communication of epidemiologic research. Our scientists work across disease areas and product types—including pharmaceuticals, medical devices, nutritional products, and environmental chemicals—to generate robust, decision-ready evidence.
Our epidemiologists and biostatisticians are equipped with the scientific and methodological expertise necessary to address the complex challenges life sciences organizations face in the ever-changing regulatory environment.
CORE AREAS OF EPIDEMIOLOGY & BIOSTATISTICS
Evidence Strategy
Define the right evidence from the start.
We evaluate the current research landscape, identify evidence gaps, and design strategic evidence generation plans that support regulatory, clinical, and commercial objectives.
Biostatistics & Advanced Analytics
Apply rigorous statistical methods to complex data.
Our biostatistics experts provide study design support, statistical analysis plans, modeling, and data visualization to generate credible, interpretable insights.
Evidence Synthesis
Synthesize existing research into actionable insights.
Our team conducts targeted and systematic literature reviews, meta-analyses, and critical appraisals of evidence (e.g., GRADE) to inform development strategy and regulatory submissions.
Clinical Trial Support
Provide the statistical foundation of clinical research.
We support clinical development programs with statistical analysis plans, trial analyses, clinical study reports, and Data Safety Monitoring Board support.
Real-World Evidence Generation
Use RWD to provide decision-ready evidence.
We design and execute epidemiologic studies using real-world data sources to evaluate treatment patterns, outcomes, safety, and effectiveness.
Scientific & Regulatory Communication
Translate evidence into clear scientific communication.
We prepare regulatory documents and responses to regulatory comments, perform medical writing, present at scientific conferences, and publish in peer-reviewed journals.
EPIDEMIOLOGY ACROSS THE PRODUCT LIFECYCLE
Epidemiology and biostatistics inform decision-making throughout product development and post-market evaluation. BRLS supports organizations at every stage of the product lifecycle.
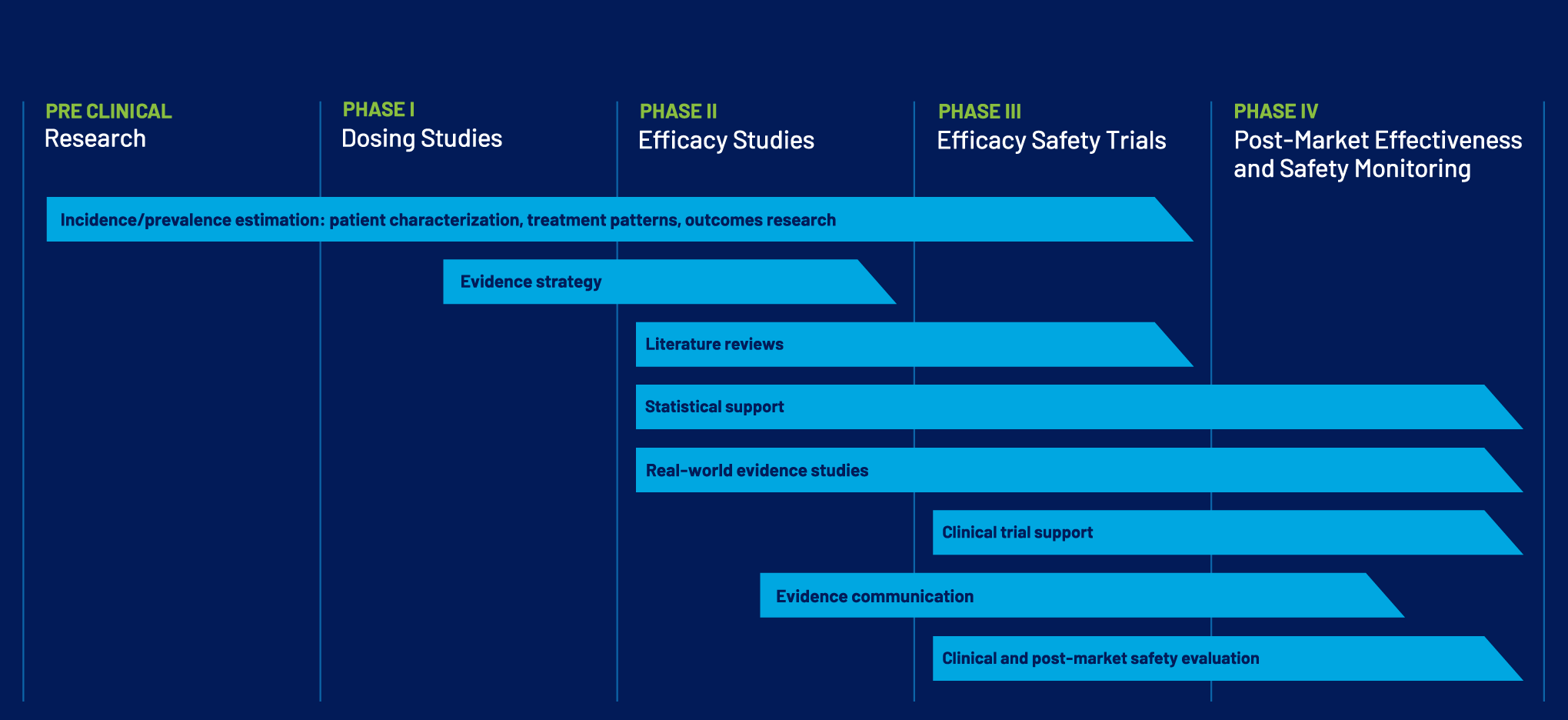
HOW WE DELIVER EVIDENCE-DRIVEN INSIGHTS
BRLS integrates epidemiology, statistics, and clinical trial and real-world data expertise to deliver scientifically rigorous insights tailored to client needs.
Deliverables may include:
Evidence strategy frameworks
Feasibility assessments
Real-world evidence study reports
Statistical analysis plans and modeling results
Data visualizations
Scientific publications and presentations
Engagement models may include:
Technical consultations
Evidence planning
Programmatic management/support
Study design and execution support
Embedded statistical or epidemiology expertise
Advisory services across development programs

CLIENTS WE SERVE
Our epidemiology and biostatistics services support organizations developing and evaluating regulated life sciences products.
-
Pharmaceutical companies
Medical device manufacturers
Nutritional and dietary product developers
Environmental and occupational health organizations
-
Clinical development teams
Epidemiology and real-world evidence groups
Biostatistics teams
HEOR
Medical affairs
Safety and pharmacovigilance leaders
Commercialization and market access teams
Generate Evidence with Confidence
Connect with BlueRidge Life Sciences to learn how our epidemiology and biostatistics experts can help you generate, analyze, and communicate evidence that supports confident scientific and regulatory decisions.
